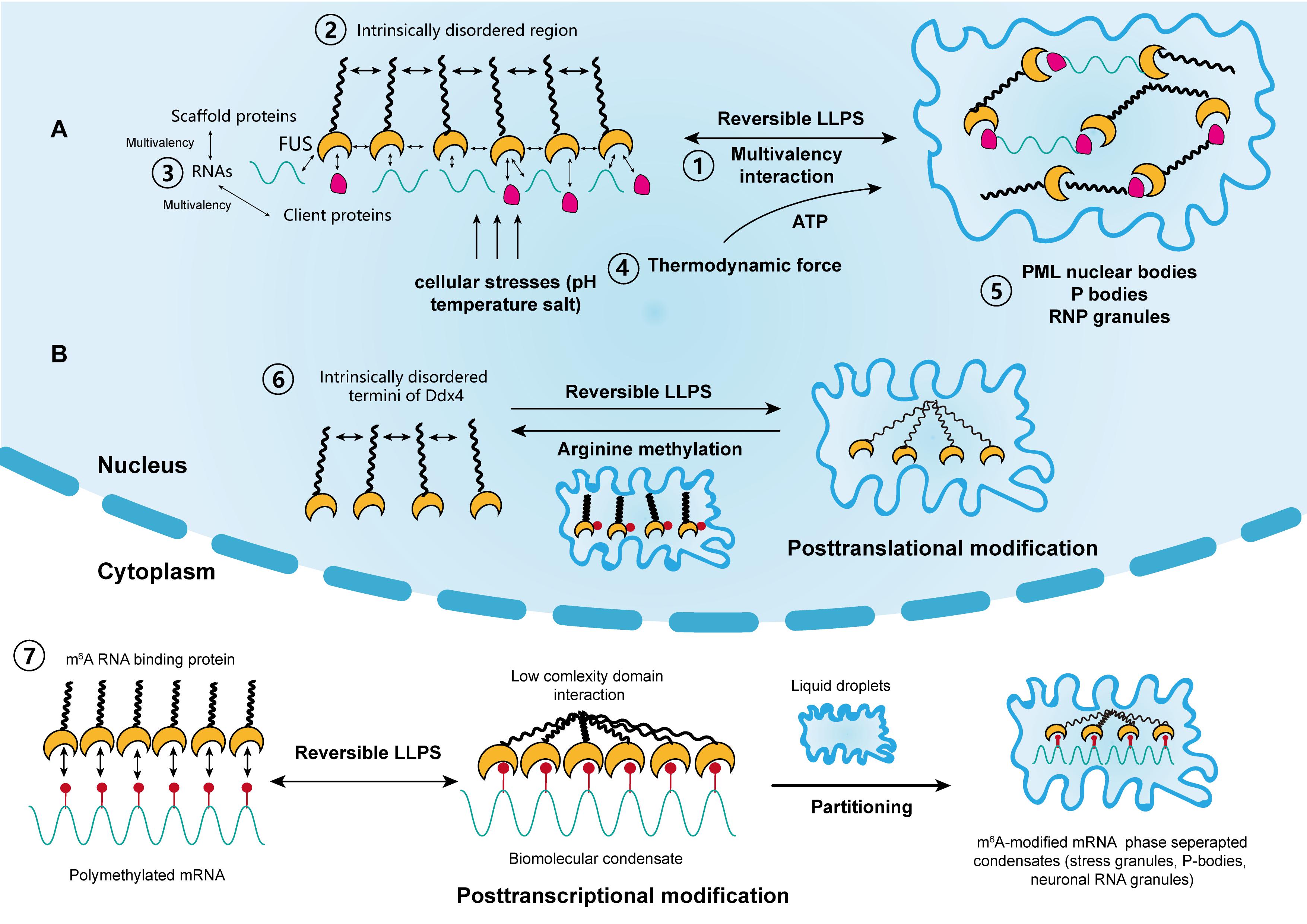
This frameshift leads to the loss of most of the RGG3 domain, the complete deletion of the NLS, and the formation of a unique C-terminal peptide sequence (fig. To investigate the consequences of ALS-causative mutations on FUS cytoplasmic function, we used the ∆14 FUS knockin mouse model ( 13), in which a mutation causing aggressive and early-onset ALS ( 14) leads to skipping of exon 14 and a frameshift in exon 15. Potential disease mechanisms have been identified in various overexpression systems ( 7, 11), although it remains unclear how well these model the physiological setting, where the expression of FUS, as well as most RBPs, is finely tuned both by auto- and cross-regulatory mechanisms ( 12). Moreover, decreased interaction with the nuclear import factor and chaperone TNPO1, posttranslational modifications and intrinsic properties given by ALS mutations all contribute to the formation of cytoplasmic FUS condensates ( 6, 7, 9, 10), the biology and composition of which are, to date, poorly understood. Under physiological conditions, the propensity of FUS to undergo LLPS in the cytoplasm is limited by its low concentration in contrast, the increased cytoplasmic localization of FUS mutants favors the transition to a phase-separated state. The role of FUS in the cytoplasm and the functional consequences of ALS-linked mutations are poorly understood however, an increasing number of studies suggest that FUS liquid-liquid phase separation (LLPS) properties play a crucial role in its cytoplasmic gain of function ( 6– 8). The loss of FUS nuclear RNA processing functions, however, is not sufficient to induce neurodegeneration on its own, drawing attention to the cytoplasmic roles of FUS and how these are affected by its increased cytoplasmic levels, which ultimately result in cytoplasmic neuronal aggregates in patient post mortem tissue ( 4, 5). Most FUS mutations disrupt the C-terminal nuclear localization signal (NLS), leading to a nuclear depletion and a cytoplasmic mislocalization of FUS ( 3). FUS mutations are causative of amyotrophic lateral sclerosis (ALS), a relentless neurodegenerative disorder in which the loss of motor neurons (MNs) leads to a progressive impairment of the neuromuscular system. Under physiological conditions, FUS is predominantly localized in the nucleus where it is involved in transcription, mRNA processing, and miRNA biogenesis ( 1) however, low levels of the protein are also present in the cytoplasm ( 2). Our results unravel new pathomechanisms of FUS-ALS and identify a novel paradigm by which mutations in one RBP favor the formation of condensates sequestering other RBPs, affecting crucial biological functions, such as protein translation.įUsed in Sarcoma (FUS) is an LCR-containing RBP involved in various aspects of RNA metabolism. Last, we show that translation of FMRP-bound RNAs is reduced in vivo in FUS-ALS motor neurons. This leads to repression of translation in mouse and human FUS-ALS motor neurons and is corroborated in vitro, where FUS and FMRP copartition and repress translation. We show that in axons, mutant FUS condensates sequester and promote the phase separation of fragile X mental retardation protein (FMRP), another RBP associated with neurodegeneration. Here, we use mouse and human models with endogenous ALS-associated mutations to study the early consequences of increased cytoplasmic FUS. FUS mutations lead to its cytoplasmic mislocalization and cause the neurodegenerative disease amyotrophic lateral sclerosis (ALS). Forman-Kay, Giampietro Schiavo, and Pietro Fratta Show FewerįUsed in Sarcoma (FUS) is a multifunctional RNA binding protein (RBP). Fisher, Alessandro Rosa, Gabriella Viero, Julie D. Nosella, Anny Devoy, Cristian Bodo, Rafaela Fernandez de la Fuente, Elizabeth M. Andrew Chong, Jack Humphrey, … Show All …, Seth Jarvis, Melis Pisiren, Oscar G. Ule, Maria Giovanna Garone, Brian Tsang, Francesca Mattedi, P.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
